Eternal questions
The search for the fundamental
The atom
Is the atom fundamental?
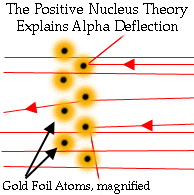
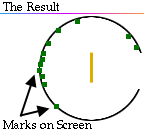
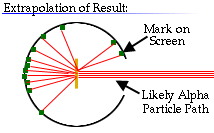
Is the nucleus fundamental?
Are protons and neutrons
fundamental? The modern atom model The scale of the atom What are we looking for? The standard model The standard model quiz
fundamental? The modern atom model The scale of the atom What are we looking for? The standard model The standard model quiz
Quarks and leptons
Matter & antimatter
What is antimatter?
Quarks
The naming of quarks
Hadrons, baryons and mesons
Leptons
Lepton decays
Lepton type conservation
Lepton decay quiz
Neutrinos
Quiz - What particles are made of
The four interactions
How does matter interact?
The unseen effect
Electromagnetism
Residual EM force
What about the nucleus?
Strong
Color charge
Quark confinement
Quarks emit gluons
Residual strong force
Weak
Electroweak
Gravity
Interaction summary
Quantum mechanics
The Pauli Exclusion Principle
Fermions and bosons
A lot to remember
End of section